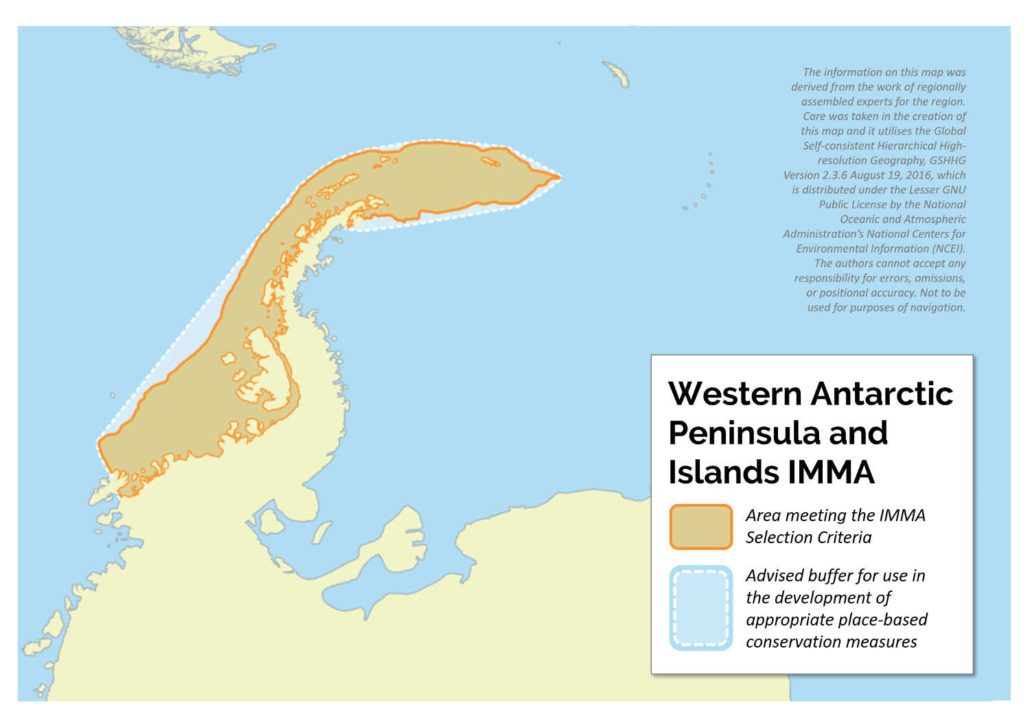
Western Antarctic Peninsula and Islands IMMA
Size in Square Kilometres
2,441,313 km2
Qualifying Species and Criteria
Humpback whale – Megaptera novaeangliae
Criterion B (2); C (2)
Fin whale – Balaenoptera physalus
Criterion A; B (2); C (2)
Antarctic minke whale – Balaenoptera bonaerensis
Criterion C (2)
Killer whale – Orcinus orca
Criterion B (1; 2); C (2); D (1)
Antarctic fur seal – Arctocephalus gazella
Criterion B (2); C (1; 2)
Southern elephant seal – Mirounga leonina
Criterion C (1; 2)
Leopard seal – Hydrurga leptonyx
Criterion B (2); C (1; 2)
Crabeater seal – Lobodon carcinophaga
Criterion B (2); C (1; 2)
Weddell seal – Leptonychotes weddellii
Criterion B (2); C (1; 2)
Marine Mammal Diversity
Criterion Dii
Balaenoptera borealis, Balaenoptera musculus intermedia, Balaenoptera acutorostrata, Eubalaena australis, Hyperoodon planifrons
Berardius arnuxii, Lagenorhynchus cruciger
Download fact sheet
Summary
The Western Antarctic Peninsula and Islands IMMA is characterized by complex bathymetry and oceanic currents that support high biological productivity. The area serves as an important feeding ground for large baleen whales, killer whales, and five species of seals. Humpback whales (Megaptera novaeangliae) are the most abundant whale species in the area and largely occupy the coastal areas on the shelf during the austral summer. In recent years Vulnerable fin whales (Balaenoptera physalus) have been observed in increasing numbers. They tend to concentrate along the shelf edge and in waters beyond the shelf, while Antarctic minke whales (Balaenoptera bonaerensis) mainly inhabit the coastal inshore waters of fjords and bays along the Antarctic Peninsula. Several killer whale (Orcinus orca) ecotypes are distributed throughout the area, with relatively high concentrations in areas such as Gerlache and Bransfield Straits. Antarctic fur seals (Arctocephalus gazella), southern elephant seals (Mirounga leonina), crabeater seals (Lobodon carcinophaga), leopard seals (Hydrurga leptonyx), and Weddell seals (Leptonychotes weddellii) reside, breed and forage in the IMMA. In addition to these abundant species, Endangered sei whales (Balaenoptera borealis) and Critically Endangered Antarctic blue whales (Balaenoptera musculus intermedia), as well as southern right whales, occasionally occur in this area.
Description of Qualifying Criteria
Criterion A – Species or Population Vulnerability
This area hosts high post-whaling densities of Vulnerable fin whales (Balaenoptera physalus) (Herr et al. 2016, Viquerat and Herr 2017). Southern Hemisphere fin whales were heavily exploited in the Southern Ocean during the commercial whaling period. Catch data suggest that they once were one of the most abundant Southern Hemisphere whale species and that they were reduced to ~2% of their pre-whaling population size (Clapham and Baker 2002). Today, the recovery status of the Southern Hemisphere fin whale population is unknown. Based on IDCR/SOWER data from surveys between 1991 and 1998, circumpolar fin whale abundance south of 60°S was last estimated at 5,445 (95 % CI 2,000–14,500) individuals (Branch and Butterworth 2001). However, this estimate almost certainly represents an unknown fraction of the total abundance of Southern Hemisphere fin whales only, as they are assumed to be extensively distributed in latitudes between 40°S and 60°S (Reilly et al. 2013) and less common south of 60°S. Recently, high sighting numbers and observations of large aggregations of fin whales in the area of the West Antarctic Peninsula have repeatedly been reported (Burkhard and Lanfredi 2012, Joiris and Dochy 2013, Santora et al. 2014; Baumann-Pickering et al. 2015; Edwards et al. 2015; Orgeira et al. 2015; Herr et al. 2016: Viquerat and Herr 2017, Seyboth 2018). In 2013, abundance of fin whales around the South Shetland Islands alone was estimated at 4,898 (95% CI 2,221–7,575) animals (Herr et al. 2016) based on an aerial line-transect survey. This estimate may be indicative of a large distributional change, with increasing numbers of fin whales moving to this area in the austral summer. Alternatively, this number is related to population recovery and increasing numbers of fin whales, returning to an area that was known for high fin whale densities during the whaling period (Kemp and Bennet 1932). Independent of these possible explanations, the number of fin whales using the shelf waters of the West Antarctic Peninsula between January and June is indisputably high and certainly represents a large fraction of the Southern Hemisphere fin whale population. The animals coming to the northern West Antarctic Peninsula in austral summer use the area as a feeding habitat, which apparently is capable of providing for a potentially recovering population and high numbers of individuals who rely on the high krill biomass of this region. Very recently, increasing use also of the inshore waters of the Bransfield Strait by fin whales has been observed (Seyboth 2018, Dalla Rosa et al. unpublished data).
Criterion B: Distribution and Abundance
Sub-criterion B1: Small and Resident Populations
The Gerlache type B killer whale (B2) is currently known only from the Western Antarctic Peninsula (Durban et al. 2016). Although all killer whale ecotypes that occur in the WAP region migrate to low latitudes, they spend relatively little time there, returning to Antarctic waters following short seasonal migrations (Durban and Pitman 2012, Pitman et al. unpublished data).
Sub-criterion B2: Aggregations
High densities of marine mammals at the West Antarctic Peninsula are supported by the extremely high krill biomass in this area (Nicol 2006, Ducklow et al. 2007, Orgeira et al. 2015, 2017). Historical whaling records underline the importance of the area for whales, with large numbers of catch records indicating high abundances of humpback, fin and blue whales before over-exploitation by commercial whaling (Kemp and Bennett 1932). Fin whales regularly aggregate in the shelf waters along the continental shelf break around the South Shetland Islands, Elephant Island and the South Orkney Islands between January and June (Burkhard and Lanfredi 2012; Joiris and Dochy 2013; Santora et al. 2014; Reyes et al. 2014, 2015; Baumann-Pickering et al. 2015; Edwards et al. 2015; Orgeira et al. 2015; Herr et al. 2016, Viquerat and Herr 2017, Seyboth 2018). As described for criterion A, abundance of fin whales is high and has to be considered to represent a considerable share of the Southern Hemisphere population. Densities have been estimated at 0.0268 ± 0.0183 ind. km−2 around Elephant Island, and at 0.0588 ± 0.0381 ind. km−2 around the South Orkney Islands based on a shipboard visual survey in 2016 (Viquerat and Herr 2016). A ship-based helicopter survey estimated fin whale density at the South Shetland Islands at 0.117 (0.053–0.181) ind. km-2 (Herr et al. 2016). The high occurrence of fin whales in the IMMA has been related to krill distribution and favourable oceanographic parameters (Santora et al. 2014, Herr et al. 2016, Viquerat et al. 2017, Seyboth 2018).
Humpback whales occur in high densities throughout the inshore waters of the area, in particular in areas such as the Bransfield Strait, the Gerlache Strait and its surrounding bays, as well as other straits and bays further south, including Marguerite Bay (Secchi et al. 2001, 2011, Friedlaender et al. 2006, 2011, Dalla Rosa 2010, Dalla Rosa et al. 2012, Nowacek et al. 2011, Johnston et al. 2012). The humpback whales migrating to this area belong to Breeding Stock G (Stevick et al. 2004, Acevedo et al. 2017). Encounter rates are available for Bransfield and Gerlache Straits (Secchi et al. 2001, 2011). Abundance has been estimated for just a portion of Bransfield Strait at 865 (95% CI 656 -1,141) individuals in 2006 (Secchi et al. 2011, Fig. 6) and at 3,024 (95%CI 944–5105) for the Bransfield Strait and adjacent areas in 2013 (Herr et al. 2016).
High encounter rates of killer whales, especially Gerlache type B, have been registered in the Gerlache Strait, although they may be found regularly throughout the West Antarctic Peninsula (Pitman and Ensor 2003, Dalla Rosa et al. 2007, unpublished data). Tracking data has revealed the importance, during post mating dispersal, of the South Orkneys and Antarctic Peninsula regions to the male Antarctic fur seals that breed on South Georgia. These male seals have been tracked as far south as Adelaide Island where numbers of males hauling out have increased over the last three decades (Lowther et al. unpublished). The connectivity between the largest breeding population, which represents 95% of the world population, and the South Orkney Islands is evidenced by the build-up of large numbers of males after the December mating season (Carlini 2006). This movement by nearly 50% of the world’s population of this species represents a significant immigration to and use of this region. The accessible beaches and pack-ice in this region provides important substrate for these animals to haul-out to rest, thermoregulate and avoid predation. Additionally, female Antarctic fur seals from Cape Shirreff are central place foragers in the region during the summer months (December-April, Hinke et al. 2017, Goebel et al. 2000). Females tracked from this site during the non-breeding period also utilise the Marguerite Bay region in April/May prior to migrating further north (Arthur et al. 2017, 2018). Approximately 15% of the crabeater, 15% of leopard and 24% of the Weddell seal population (from surveyed areas) reside, breed and forage in the Western Antarctic Peninsula (Southwell et al. 2012).
Criterion C: Key Life Cycle Activities
Sub-criterion C1: Reproductive Areas
The Western Antarctic Peninsula region encompassing the IMMA is a breeding area for a proportion of crabeater, leopard and Weddell seal populations (Southwell et al. 2012). Also, the most southerly Antarctic fur seal and southern elephant seal breeding colonies are located in the South Shetland Islands and Elephant Island (Le Boeuf and Laws 1994, Goebel et al. 2000).
Sub-criterion C2: Feeding Areas
The Western Antarctic Peninsula region is characterized by a complex bathymetry and circulation, which favours enhanced biological productivity, influencing the distribution and abundance of cetaceans (e.g. Friedlaender et al. 2006, Dalla Rosa 2010). The area is known for extremely high krill biomass (Nicol 2006) supporting high predator densities. The area serves as a feeding ground for several baleen whale species, which migrate from their breeding grounds in more temperate waters to feed on krill during the austral summer. A variety of sympatric krill predators has been shown to occur in the area (Friedlaender et al. 2008, Santora and Veit 2013). Satellite tracking of humpback (Dalla Rosa et al. 2008; Curtice et al. 2015, Weinstein and Friedlaender 2017) and fin whales (Dalla Rosa et al. unpublished data) has shown that individuals consistently use this area for extended periods of time during the feeding season. Feeding behaviour has been described for humpback whales by means of telemetry (Friedlaender et al. 2013). Visual observations of bubble nets and other observed feeding behaviour prove feeding of humpback and fin whales in the area (Acevedo et al. 2008, Herr et al. 2016). Large aggregations of up to 60 surface feeding fin and humpback whales have been observed in the proposed area, preying on krill (Nowacek et al. 2011, Burkhardt and Lanfredi 2012, Joiris and Dochy 2013, Herr et al. 2016). Their distribution has been proposed to be linked to high densities of krill (Santora et al. 2014, Herr et al 2016). For humpback whales the area encompasses the major part of the feeding grounds of Breeding Stock G (Stevick et al. 2004, Dalla Rosa et al. 2012, Acevedo et al. 2017).
All Antarctic killer whale ecotypes also use this area for feeding, as differences in foraging ecology or prey preferences have helped to distinguish among them (e.g. Durban et al. 2016). Approximately 15% of the crabeater seal population (from surveyed areas) reside, breed and forage in the WAP (Southwell et al. 2012). The majority of crabeater seal feeding dives occur within the top 50 m, although they can dive up to 600 m with a maximum duration of 24 minutes (Burns et al. 2004, 2008). They appear to favour foraging locations on the continental shelf within the 50 to 450 m depth range, with a tendency to avoid depths of 600 m or greater. Crabeater Seals feed primarily on Antarctic Krill (Euphausia superba), which accounts for over 90% of their diet, with the remainder made up of fish and squid. Deeper dives at dawn and dusk indicate that their feeding activity is tied to the daily vertical migrations of Krill. Daytime foraging appears to exploit zooplankton schools compressed along the bottom. Their high density within the Western Antarctic Peninsula is likely related to the abundance of krill in this region. Winter tracking data suggests that they alter their behaviour to accommodate seasonal and/or annual fluctuations in seasonal sea ice and associate with bathymetric features likely to concentrate prey patches (Burns et al. 2004). They are most likely located in nearshore waters where bathymetric gradients and ice concentration are high (Burns et al. 2004).
The Western Antarctic Peninsula is home to 24% of the Weddell seal population (from surveyed areas, Southwell et al. 2012). The abundance of prey and availability of suitable sea-ice habitat makes this area ideal for this pagophilic species. Weddell seals are generalist predators, and their diet likely varies at a regional scale. In the Western Antarctic Peninsula they are known to feed on Antarctic krill, notothenioid fish (icefishes), particularly the Antarctic silverfish, but the diet also includes Antarctic toothfish, myctophids and cephalopods. Weddell seals typically forage within two depth layers, surface waters (0-160m) and near the bottom. While both pelagic and benthic diving occur during daylight, seals forage almost exclusively in the upper water column at night (Plötz et al. 2001). Leopard seals have a circumpolar distribution concentrated in, but not limited to, the Southern Ocean. They are most commonly found in and around the outer fringes of the pack ice or close to the Antarctic Continent. The Western Antarctic Peninsula represents 15% of the surveyed populations (Southwell et al. 2012). They are top predators in the Antarctic marine ecosystem and feed on a diverse range of prey that varies seasonally, spatially and ontogenically including krill, fish, squid, penguins and other pinnipeds. Penguins are eaten throughout the year but form an especially large proportion of the leopard seal diet in January to March, when these birds are breeding (Siniff and Stone 1985). Newly weaned crabeater seal pups can also be an important part of the diet in the pack ice near the Antarctic Peninsula (Siniff and Stone 1985). Leopard seal predation has been implicated in slowing the recovery of the Antarctic fur seal population at Elephant Island (Boveng et al. 1998). At Cape Shirreff, studies monitoring leopard seal predation on Antarctic fur seal pups suggest that they consume up to half of the annual pup production within this area. In addition to fur seal pups and penguins, leopard seals were found to take two species of demersal fish (Gobionotothen gibberifrons and Notothenia coriiceps) and scavenge carcasses of fur seals and penguins.
Male Antarctic fur seals from South Georgia have been tracked traveling to and moving within the South Orkney Islands and Western Antarctic Peninsula region (Boyd et al. 1998, BAS unpublished). This southwards migration of a large predator biomass has significant implications for consumption of krill, fish and penguins within the region (Davis et al. 2006). The growing body of at sea movement data shows that male fur seals at South Georgia display some level of central place foraging alternating time at sea with periods ashore resting, very similar to lactating females. These seals also predominantly foraged on-shelf, close to their haul out locations (<100 km), and so have a greater spatial overlap with penguins than the more pelagic foraging female seals. Post breeding males often have very poor body condition resulting from their prolonged fast whilst competing for territory and potential mates. The abundant food resources in the South Orkney Islands and the Western Antarctic Peninsula, in close proximity to accessible haul-out locations makes this an ideal area for these predators. The post-breeding foraging areas of southern elephant seals are close to their breeding grounds for both males and females (Huckstadt et al. 2012; Muelbert et al. 2013, Hindell et al. 2016). They feed along the continental shelf and in the trough areas, and satellite tracking shows they range throughout the entire area the IMMA. Criterion D: Special Attributes
Sub-criterion D1: Distinctiveness
Though three killer whale ecotypes frequently occur in this area (Pitman and Ensor 2003, Dalla Rosa et al. 2007, Durban et al. 2016), this area is particularly important for Gerlache type B ecotype, as these are only found around the Antarctic Peninsula (Durban et al. 2016).
Sub-criterion D2: Diversity
Species richness and abundance of seabirds and marine mammals and areas that are persistently attractive to top predators have been demonstrated for the proposed area (Lazaneo et al. 2013, Santora and Veit 2013). At least 9 marine mammal species occur regularly in the proposed area (humpback, fin, Antarctic minke and killer whales, Antarctic fur, leopard, Weddell, crabeater and elephant seals). The highest cetacean diversity has been recorded in the northern Antarctic Peninsula, including the area around Elephant Island (e.g. Lazaneo et al. 2013). Additional records of marine mammals throughout the proposed area include encounters of sei whales (Balaenoptera borealis) (Secchi et al. 2001), dwarf minke whales (Balaenoptera acutorostrata) (Acevedo et al. 2011), Antarctic blue whales (Balaenoptera musculus intermedia) (Herr et al. 2016) and southern right whales (Eubalaena australis) (Stone and Hammer 1988, Aguayo-Lobo et al. 2008, Johnston et al. 2012, Dalla Rosa, unpublished data, Herr et al. unpublished data). Odontocete species that have been reported from the proposed area include hourglass dolphins (Lagenorhynchus cruciger) (Santora 2012, Dellabianca et al. 2012), southern bottlenose whales (Hyperoodon planifrons) (Santora and Brown 2010, Herr et al. unpublished data) and Arnoux’s beaked whales (Berardius arnuxii) (Johnston et al. 2012, Friedlaender et al. 2010, Herr et al. unpublished data).
Supporting Information
Acevedo, J., Aguayo-Lobo, A. and Plana, J. 2008. ‘Conducta de Alimentación de la Ballena Jorobada en la Península Antártica’. Boletín Antártico Chileno, 27(1):21-22.
Acevedo, J., Olavarría, C., Plana, J., Aguayo-Lobo, A., Larrea, A. and Pastene, L.A. 2011. ‘Occurrence of Dwarf Minke Whales (Balaenoptera acutorostrata subsp.) Around the Antarctic Peninsula’. Polar Biology, 34:313-318.
Acevedo, J., Aguayo-Lobo, A., Allen, J., Botero-Acosta, N., Capella, J., Castro, C., Dalla Rosa, L., Denkinger, J., Félix, F., Flórez-González, L., Garita, F., Guzmán, H.M., Haase, B., Kaufman, G., Llano, M., Olavarría, C., Pacheco, A.S., Plana, J., Rasmussen, K., Scheidat, M., Secchi, E.R., Silva, S. and Stevick, P.T. 2017. ‘Migratory Preferences of Humpback Whales between Feeding and Breeding Grounds in the Eastern South Pacific’. Marine Mammal Science, 33:1035-1052.
Aguayo-Lobo A., Acevedo, J, Brito, J.L., Olavarría, C., Moraga, R. and Olave, C. 2008. ‘La Ballena Franca del Sur, Eubalaena australis (Desmoulins, 1822) en Águas Chilenas: Análisis de sus Registros desde 1976 a 2008’. Revista de Biología Marina y Oceanografía, 43(3):653-668. DOI: 10.4067/S0718-19572008000300024
Arthur, B., Hindell, M., Bester, M., Trathan, P., Jonsen, I., Staniland, I., Oosthuizen, W.C., Wege, M., Lea, M.A. 2015. ‘Return customers: Foraging site fidelity and the effect of environmental variability in wide-ranging Antarctic fur seals‘. PloS one, 10(3):e0120888.
Arthur, B., Hindell, M., Bester, M.N., Oosthuizen, W.C., Wege, M and Lea, M.A. 2016. South for the Winter? Within‐dive Foraging Effort Reveals the Trade‐offs between Divergent Foraging Strategies in a Free‐ranging Predator’. Functional Ecology, 10:1623-37.
Arthur, B., Hindell, M., Bester, M., De Bruyn, P.J.N., Trathan, P., Goebel, M. and Lea, M.A. 2017. ‘Winter Habitat Predictions of a Key Southern Ocean Predator, the Antarctic Fur Seal (Arctocephalus gazella)’. Deep Sea Research Part II: Topical Studies in Oceanography, 140:171-81.
Arthur, B., Hindell, M., Bester, M., De Bruyn, P.J.N., Goebel, M.E., Trathan, P., Lea, M.A. 2018. ‘Managing for change: Using vertebrate at sea habitat use to direct management efforts’. Ecological Indicators, 91:338-49.
Bassoi, M., Acevedo, J., Secchi, E.R., Aguayo-Lobo, A., Dalla Rosa, L., Torres, D., Santos, M.C.O. and Azevedo, A.F. 2020. ‘Cetacean distribution in relation to environmental parameters between Drake Passage and northern Antarctic Peninsula’. Polar Biology, 43:1-15. DOI: 10.1007/s00300-019-02607-z
Baumann-Pickering, S., Širović, A., Trickey, J.S., Hildebrand, J.A., Reyes Reyes, V., Melcón, M.L. and Iñíguez, M.A. 2015. ‘Cetacean Presence near Elephant Island, Antarctica, Based on Passive Acoustic Monitoring’, paper SC/66a/SH/18 delivered to the IWC Scientific Committee, May 2015.
Bengtson, J.L. and Stewart, B.S. 1992. ‘Diving and Haulout Behavior of Crabeater Seals in the Weddell Sea, Antarctica during March 1986’. Polar Biology, 12:635-644.
Boyd, I.L., McCafferty, D.J., Reid, K., Taylor, R. and Walker, T.R. 1998. ‘Dispersal of Male and Female Antarctic Fur Seals (Arctocephalus gazella)’. Canadian Journal of Fisheries and Aquatic Sciences, 55(4):845-852.
Boyd, I.L., Staniland, I.J. and Martin, A.R. 2002. ‘Distribution of foraging by female Antarctic fur seals’. Marine Ecology Progress Series, 242:285-294.
Boveng, P.L., Hiruki, L.M., Schwartz, M.K. and Bengtson, J.L. 1998. ‘Population Growth of Antarctic Fur Seals: Limitation by a Top Predator, the Leopard Seal?’. Ecology, 79(8):2863-2877.
Brierley, A.S., Fernandes, P.G., Brandon, M.A., Armstrong, F., Millard, N.W., McPail, S.D., Stevenson, P., Pebody, M., Perret, J., Squires, M., Bone, D.G. and Griffiths, G. 2002. ‘Antarctic Krill Under Sea Ice: Elevated Abundance in a Narrow Band just South of Ice Edge’. Science, 295:1890-1892.
Brierley, A.S., Watkins, J.L., Goss, C., Wilkinson, M.T. and Everson, I. 1999. ‘Acoustic Estimates of Krill Density at South Georgia, 1981 to 1998’. CCAMLR Science, 6:47-57.
Burns, J.M., Costa, D.P., Fedak, M.A., Hindell, M.A., Bradshaw, C.J., Gales, N.J. and Crocker, D.E. 2004. ‘Winter Habitat Use and Foraging Behavior of Crabeater Seals Along the Western Antarctic Peninsula’. Deep Sea Research Part II: Topical Studies in Oceanography, 51(17): 2279-2303.
Burns, J.M., Hindell, M.A., Bradshaw, C.J.A. and Costa, D.P. 2008. ‘Fine-scale Habitat Selection of Crabeater Seals as Determined by Diving Behavior’. Deep Sea Research Part II: Topical Studies in Oceanography, 55(3):500-514.
Burkhard, E. and Lanfredi, C. 2012. ‘Fall Feeding Aggregations of Fin Whales off Elephant Island (Antarctica)’, paper SC/64/SH9 delivered to the IWC Scientific Committee (unpublished). http://epic.awi.de/30452/1/SC-64-SH9_corrected_may22.pdf
Branch, T.A. and Butterworth, D.S. 2001. ‘Estimates of Abundance South of 60°S for Cetacean Species Sighted Frequently on the 1978/79 to 1997/98 IWC/IDCR-SOWER Sighting Surveys’. Journal of Cetacean Research and Management, 3:251−270.
Carlini, A.R., Daneri, G.A., Casaux, R. and Márquez, M.E. 2006. ‘Haul‐out Pattern of Itinerant Male Antarctic Fur Seals (Arctocephalus gazella) at Laurie Island, South Orkney Islands’. Polar Research, 25:139-144.
Carrick, R., Csordas, S.E. and Ingham, S.E. 1962. ‘Studies on the Southern elephant seal, Mirounga leonine (L.). IV. Breeding and development’. C.S.LR.O. Wildlife Research, 7: 161-197.
Clapham, P.J. and Baker, C.S. 2002. ‘Modern Whaling’. In: W.F. Perrin, B. Würsig and J.G.M.Thewissen (eds.) Encyclopedia of Marine Mammals, pp. 1328–1332. New York: Academic Press.
Costa, D.P., Huckstadt, L.A., Crocker, D.E., McDonald, B.I., Goebel, M.E. and Fedak, M.A. 2010. ‘Approaches to studying climatic change and its role on the habitat selection of Antarctic pinnipeds’. Integrative and Comparative Biology, 50(6):1018-1030.
Curtice, C., Johnston, D.W., Ducklow, H., Gales, N., Halpin, P.N. and Friedlaender, A.S. 2015. ‘Modeling the Spatial and Temporal Dynamics of Foraging Movements of Humpback Whales (Megaptera novaeangliae) in the Western Antarctic Peninsula’. Movement Ecology, 3:13. DOI 10.1186/s40462-015-0041-x
Dalla Rosa, L., Bassoi, M., Secchi, E.R., Danilewicz, D.S., Moreno, I.B., Santos, M.C.O. and Flores, P.A.C. 2007. ‘Occurrence and Distribution of Killer Whales in Waters of the Antarctic Peninsula’, paper SC/59/SM10 delivered to the Scientific Committee Meeting of the International Whaling Commission (IWC), Anchorage, Alaska, USA.
Dalla Rosa, L., Secchi, E.R., Maia, Y.G., Zerbini, A.N. and Heide-Jørgensen, M.P. 2008. ‘Movements of Satellite-monitored Humpback Whales on Their Feeding Ground Along the Antarctic Peninsula’. Polar Biology, 31:771–781.
Dalla Rosa, L., Félix, F., Stevick, P., Secchi, E.R., Allen, J.M., Chater. K., Martin, A.R. and Bassoi, M. 2012. ‘Feeding Grounds of the Eastern South Pacific Humpback Whale Population Include the South Orkney Islands’. Polar Research, 31:17324.
Dalla Rosa, L. 2010. ‘Modeling the Foraging Habitat of Humpback Whales’. PhD thesis. Vancouver: The University of British Columbia.
Davis, D., Staniland, I.J. and Reid, K. 2006. ‘Spatial and Temporal Variability in the Fish Diet of Antarctic Fur Seal (Arctocephalus gazella) in the Atlantic Sector of the Southern Ocean’. Canadian Journal of Zoology, 84(7):1025-1037.
Dellabianca, N., Scioscia, G., Schiavini, A. and Raya Rey, A. 2012. ‘Occurrence of Hourglass Dolphin (Lagenorhynchus cruciger) and Habitat Characteristics along the Patagonian Shelf and the Atlantic Ocean Sector of the Southern Ocean’. Polar Biology, 35:1921-1927. doi:10.1007/s00300-012-1217-0
Ducklow, H.W., Baker, K., Fraser, W.R., Martinson, D.G., Quetin, L.B., Ross, R.M., Smith, R.C., Stammerjohn, S. and Vernet, M. 2007. ‘Marine Pelagic Ecosystems: the West Antarctic Peninsula’. Philosophical Transactions of the Royal Society B, 362:67-94.
Durban, J.W. and Pitman, R.L. 2012. ‘Antarctic Killer Whales Make Rapid, Round-trip Movements to Subtropical Waters: Evidence for Physiological Maintenance Migrations?’. Biology Letters, 8:274-277.
Durban, J.W., Fearnbach, H., Burrows, D.G., Ylitalo, G.M. and Pitman, R.L. 2016. ‘Morphological and Ecological Evidence for Two Sympatric Forms of Type B Killer Whale Around the Antarctic Peninsula’. Polar Biology, 40(1):231-236.
Edwards, E.F., Hall, C., Moore, T.J., Sheredy, C. and Redfern, J.V. 2015. ‘Global Distribution of Fin Whales Balaenoptera physalus in the Post-whaling Era (1980-2012)’. Mammal Review, 45:197-214.
Forcada, J. and Staniland I.J. 2018. Antarctic Fur Seal: Arctocephalus gazella. In: B. Würsig, J.G.M. Thewissen K.M. Kovacs (eds), Encyclopedia of Marine Mammals (Third Edition), pp. 25-27. Academic Press.
Friedlaender, A.S., Halpin, P.N., Qian, S.S., Lawson, G.L., Wiebe, P.H., Thiele, D. and Read, A.J. 2006. ‘Whale Distribution in Relation to Prey Abundance and Oceanographic Processes in Shelf Waters of the Western Antarctic Peninsula’. Marine Ecology Progress Series, 317:297-310.
Friedlaender, A.S., Nowacek, D.P., Johnston, D.W., Read, A.J., Tyson, R.B., Peavey, L. and Revelli, E.M.S. 2010. ‘Multiple Sightings of Large Groups of Arnoux’s Beaked Whales (Berardius arnouxii) in the Gerlache Strait, Antarctica’. Marine Mammal Science, 26(1):246-250.
Friedlaender, A.S., Johnston, D.W., Fraser, W.R., Burns, J., Halpin, P.N. and Costa, D.P. 2011. ‘Ecological Niche Modelling of Sympatric Krill Predators Around Marguerite Bay, Western Antarctic Peninsula’. Deep-Sea Research Part II, 58:1729–1740.
Friedlaender, A.S., Tyson, R.B., Stimpert, A.K., Read, A.J. and Nowacek, D.P. 2013. ‘Extreme Diel Variation in the Feeding Behavior of Humpback Whales along the Western Antarctic Peninsula During Autumn’. Marine Ecology Progress Series, 494:281-289.
Friedlaender, A.S., Goldbogen, J.A., Nowacek, D.P., Read, A.J., Johnston, D. and Gales, N. 2014. ‘Feeding Rates and Under-ice Foraging Strategies of the Smallest Lunge Filter Feeder, the Antarctic Minke Whale (Balaenoptera bonaerensis)’. Journal of Experimental Biology, 217:2851-2854.
Goebel, M.E. 2002. ‘Northern Fur Seal Lactation, Attendance and Reproductive Success in Two Years of Contrasting Oceanography’. Ph.D. Thesis. University of California.
Goebel, M.E., Costa, D.P., Crocker, D.E., Sterling, J.E. and Demer, D.A. 2000. ‘Foraging Ranges and Dive Patterns in Relation to Bathymetry and Time-of-day of Antarctic Fur Seals, Cape Shirreff, Livingston Island Antarctica. In: W. Davisons, C. Howard-Williams and P. Broady (eds.) Antarctic Ecosystems: Models for Wider Ecological Understanding, pp.47-50. Christchurch: New Zealand Natural Sciences Press.
Goebel, M.E., Krause, D., Freeman, S., Burner, R., Bonin, C., Vasquez del Mercado, R., Van Cise, A.M. and Gafney, J. 2008. Pinniped Research at Cape Shirreff, Livingston Island, Antarctica, 2008/09.
Goebel, M.E., Perryman, W.L., Hinke, J.T., Krause, D.J., Hann, N.A., Gardner, S. and LeRoi, D.J. 2015. ‘A small unmanned aerial system for estimating abundance and size of Antarctic predators’. Polar Biology, 38(5):619-630.
Herr, H., Viquerat, S., Siegel, V., Kock, K.H., Dorschel, B., Huneke, W.G.C., Bracher, A., Schröder, M. and Gutt, J. 2016. ‘Horizontal Niche Partitioning of Humpback and Fin Whales Around the West Antarctic Peninsula – Evidence from a Concurrent Whale and Krill Survey’. Polar Biology, 39:799- 818.
Herr, H., Kelly, N., Dorschel, B., Huntemann, M., Kock, K., Lehnert, L.S., Siebert, U., Viquerat, S., William, R. and Scheidat, M. 2019. ‘Aerial surveys for Antarctic minke whales (Balaenoptera bonaerensis) reveal sea ice dependent distribution patterns’. Ecology and Evolution, 9(10):5664-5682. https://doi.org/10.1002/ece3.5149
Hindell, M.A., McMahon, C.R., Bester, M.N., Boehme, L., Costa, D., Fedak, M.A., Guinet, C., Herraiz‐Borreguero, L., Harcourt, R.G., Huckstadt, L., Kovacs, K.M., Lydersen C., McIntyre, T., Muelbert, M., Patterson, T., Roquet, F., Williams, G. and Charrassin, J.B. 2016. ‘Circumpolar Habitat Use in the Southern Elephant Seal: Implications for Foraging Success and Population Trajectories’. Ecosphere, 7(5):e01213.
Hindell, M.A., Reisinger, R.R., Ropert-Coudert, Y., Huckstadt, L.A., Trathan, P.N., Bornemann, H., Charrassin, J.-B., Chown, S.L., Costa, D.P., Danis, B., Lea, M.-A., Thompson, D., Torres, L.G., Van de Putte, A.P., Alderman, R., Andrews-Goff, V., Arthur, B., Ballard, G., Bengtson, J., Bester, M.N., Blix, A.S., Boehme, L., Bost, C.-A., Boveng, P., Cleeland, J., Constantine, R., Corney, S., Crawford, R.J.M., Dalla Rosa, L., de Bruyn, P.J.N., Delord, K., Descamps, S., Double, M., Emmerson, L., Fedak, M., Friedlaender, A., Gales, N., Goebel, M.E., Goetz, K.T., Guinet, C., Goldsworthy, S.D., Harcourt, R., Hinke, J.T., Jerosch, K., Kato, A., Kerry, K.R., Kirkwood, R., Kooyman, G.L., Kovacs, K.M., Lawton, K., Lowther, A.D., Lydersen, C., Lyver, P., Makhado, A.B., Marquez, M.E.I., Mcdonald, B.I, McMahon, C.R., Muelbert, M., Nachtsheim, D., Nicholls, K.W., Nordøy, E.S., Olmastroni, S., Phillips, R.A., Pistorius, P., Plötz, J., Pütz, K., Ratcliffe, N., Ryan, P.G., Santos, M., Southwell, C., Staniland, I., Takahashi, A., Tarroux, A., Trivelpiece, W., Wakefield, E., Weimerskirch, H., Wienecke, B., Xavier, J.C., Wotherspoon, S. Jonsen, I.D. and Raymond, B. 2020. ‘Tracking of marine predators to protect Southern Ocean ecosystems’. Nature, 580: 87–92. DOI: https://www.nature.com/articles/s41586-020-2126-y
Hinke, J.T., Cossio, A.M., Goebel, M.E., Reiss, C.S., Trivelpiece, W.Z. and Watters, G.M. 2017. ‘Identifying Risk: Concurrent Overlap of the Antarctic Krill Fishery with Krill-Dependent Predators in the Scotia Sea’. PLOS ONE, 12(1):e0170132.
Hofmann, E.E. and Murphy, E.J. 2004. ‘Advection, Krill, and Antarctic Marine Ecosystems’. Antarctic Science, 16(4):487-499.
Hucke-Gaete, R., Osman, L.P., Moreno, C.A. and Torres, D. 2004. ‘Examining natural population growth from near extinction: the case of the Antarctic fur seal at the South Shetlands, Antarctica’. Polar Biology, 27(5):304-311.
Hückstädt, L.A., Koch, P.L., McDonald, B.I., Goebel, M.E., Crocker, D.E. and Costa, D.P. 2012. ‘Stable Isotope Analyses Reveal Individual Variability in the Trophic Ecology of a Top Marine Predator, the Southern Elephant Seal’. Oecologia, 169:395–406.
IPCC. 2007. Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. S. Solomon, D. Qin, M. Manning, Z. Chen, M. Marquis, K.B. Averyt, M. Tignor, H.L. Miller (eds.). Cambridge: Cambridge University Press.
IPCC. 2013. Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Final Draft (7 June 2013).
Johnston, D.W., Friedlaender, A.S., Read, A.J. and Nowacek, D.P. 2012. ‘Initial Density Estimates of Humpback Whales, Megaptera novaeangliae, in the Inshore Waters of the Western Antarctic Peninsula during the Late Autumn’. Endangered Species Research, 18:63–71. DOI: 10.3354/esr00395
Joiris, C.R. and Dochy, O. 2013. ‘A Major Autumn Feeding Ground for Fin Whales, Southern Fulmars and Grey-headed Albatrosses Around the South Shetland Islands, Antarctica’. Polar Biology, 36(11):649-1658.
Kasamatsu, F., Ensor, P., Joyce, G.G. and Kimura, N. 2000. ‘Distribution of Minke Whales in the Bellingshausen and Amundsen Seas (60°W-120°W), with Special Reference to Environmental/Physiographic Variables’. Fisheries Oceanography, 9:214-223.
Kawamura, A. 1994. ‘A Review of Baleen Whale Feeding in the Southern Ocean’. Reports of the International Whaling Commission, 44:261-271.
Kemp, S. and Bennett, A.G. 1932. ‘On the Distribution and Movements of Whales on the South Georgia and South Shetland Whaling Grounds’. Discovery Reports, 6:165–190.
Krause, D.J., Goebel, M.E., Marshall, G.J. and Abernathy, K. 2015. ‘Novel foraging strategies observed in a growing leopard seal (Hydrurga leptonyx) population at Livingston Island, Antarctic Peninsula’. Animal Biotelemetry, 3(1).
Krause, D.J., Goebel, M.E., Marshall, G.J. and Abernathy, K. 2016. ‘Summer diving and haul-out behavior of leopard seals (Hydrurga leptonyx) near mesopredator breeding colonies at Livingston Island, Antarctic Peninsula’. Marine Mammal Science, 32(3):839-867.
Laws, R.M. (ed.) 2009. Antarctic Seals: Research Methods and Techniques. Cambridge University Press.
Lazaneo, C.Z., Dalla Rosa, L. and Secchi, E.R. 2013. ‘Structure of the Cetacean Community Around the Antarctic Peninsula’. Annual Activity Report 2012 of National Institute of Science and Technology – Antarctic Environmental Research (INCT-APA), pp.149-154. São Carlos: Editora Cubo.
Le Boeuf, B.J. and Laws, R.M. (eds.) 1994. Elephant Seals: Population Ecology, Behavior, and Physiology. University of California Press.
Loeb, V., Siegel, V., Holm-Hansen, O., Hewitt, R., Fraser, W., Trivelpiece, W. and Trivelpiece, S. 1997. ‘Effects of Sea-ice Extent and Krill or Salp Dominance on the Antarctic Food Web’. Nature, 387:897–900.
Mackintosh, N.A. 1970. ‘Whales and Krill in the Twentieth Century’. Antarctic Ecology, 1:195-212.
McDonald, B.I., Goebel, M.E., Crocker, D.E. and Costa, D.P. 2010. ‘Maternal investment in the Antarctic fur seal: Impacts of maternal traits, pup traits, and provisioning strategy’. Integrative and Comparative Biology, 50:E113-E113.
McDonald, B.I., Goebel, M.E., Crocker, D.E. and Costa, D.P. 2012. ‘Biological and environmental drivers of energy allocation in a dependent mammal, the Antarctic fur seal pup’. Physiological and Biochemical Zoology 85(2):134-147.
Meyer, B. and Wessels, W. 2018. ‘The Expedition PS112 of the Research Vessel POLARSTERN to the Antarctic Peninsula Region in 2018’. Reports on Polar and Marine Research, Bremerhaven, Alfred Wegener Institute for Polar and Marine Research, 722, 125 p. DOI: https://doi.org/10.2312/BzPM_0722_2018
Muelbert, M.M.C., De Souza, R.B., Lewis, M.N. and Hindell, M.A. 2013. ‘Foraging Habitats of Southern Elephant Seals, Mirounga leonina, from the Northern Antarctic Peninsula’. Deep-Sea Research Part II, (88-89):47-60.
Nicol, S. 2006. ‘Krill, Currents, and Sea Ice: Euphausia superba and Its Changing Environment’. BioScience, 56(2): 111–120.
Nicol, S., Constable, A.J. and Pauly, T. 2000. ‘Estimates of Circumpolar Abundance of Antarctic Krill Based on Recent Acoustic Density Measurements’. CCAMLR Science, 7:87–99.
Nowacek, D.P., Friedlaender, A.S., Halpin, P.N., Hazen, E.L., Johnston, D.W., Read, A.J., Espinasse, B., Zhou, M. and Zhu, Y. 2011. ‘Super-Aggregations of Krill and Humpback Whales in Wilhelmina Bay, Antarctic Peninsula’. PLOS ONE, 6(4):e19173.
Oliva, M., Navarro, F., Hrbáček, F., Hernández, A., Nývlt, D., Pereira, P., Ruiz-Fernández, J., Trigo, R. 2017. ‘Recent Regional Climate Cooling on the Antarctic Peninsula and Associated Impacts on the Cryosphere’. Science of the Total Environment, 580:210-223.
Oreskes, N. 2004. The Scientific Consensus on Climate Change’. Science, 306:1686-1686.
Orgeira, J.L., Alderete, M.C., Jiménez, Y.G. and González, J.C. 2015. ‘Long-Term Study of At-Sea Distribution of Seabirds and Marine Mammals in the Scotia Sea, Antartica’. Advances in Polar Science, 26:158-167.
Orgeira, J.L., González, J.C., Jiménez, Y.G., Benítez, L.M. 2017. ‘Occurrence of Fin and Humpback Whales in the Scotia Sea and the Protected Marine Area of the South Orkney Islands, Antarctica’. Mastozoología Neotropical, 24(1):135-143.
Osman, L.P., Hucke-Gaete, R., Moreno, C.A. and Torres D. 2004. ‘Feeding ecology of Antarctic fur seals at Cape Shirreff, South Shetlands, Antarctica‘. Polar Biology, 27(2):92-98.
Öztürk, B., Tonay, M.A., Öz, M.I., Yilmaz, I.N., Ergül, H.A. and Öztürk, A.A. 2017. ‘Sightings of Cetaceans in the Western Antarctic Peninsula during the First Joint Turkish–Ukrainian Antarctic Research Expedition’. Turk Journal Zoology, 41:955-961. DOI:10.3906/zoo-1611-48.
Paijmans, A. J., Stoffel, M.A., Bester, M.N., Cleary, A.C., De Bruyn, P.J.N., Forcada, J., Goebel, M.E., Goldsworthy, S.D., Guinet, C., Lydersen, C., Kovacs, K.M., Lowther, A. and Hoffman, J.I. 2020. ‘The genetic legacy of extreme exploitation in a polar vertebrate’. Scientific reports, 10(1):1-12.
Pitman, R.L. and Ensor, P. 2003. ‘Three Forms of Killer Whales (Orcinus orca) in Antarctic Waters’. Journal of Cetacean Research and Management, 5:131–139.
Pitman, R.L., Durban, J.W., Joyce, T., Fearnbach, H., Panigada, S. and Lauriano, G. 2020. ‘Skin in the game: epidermal molt as a driver of long‐distance migration in whales’. Marine Mammal Science, 36(2):565-594. DOI: 10.1111/mms.12661
Plötz, J., Bornemann, H., Knust, R., Schröder, A. and Bester, M. 2001. ‘Foraging Behaviour of Weddell Seals, and Its Ecological Implications’. Polar Biology, 24:901-909.
Ropert-Coudert, Y., Van de Putte, A.P., Reisinger, R., Bornemann, H., Charrassin, J.-B., Costa, D.P., Danis, B., Huckstadt, L.A., Jonsen, I.D., Lea, M.-A., Thompson, D., Torres, L.G., Trathan, P.N., Wotherspoon, S., Ainley, D.G., Alderman, R., Andrews-Goff, V., Arthur, B., Ballard, G., Bengtson, J., Bester, M.N., Blix, A.S., Boehme, L., Bost, C.-A., Boveng, P., Cleeland, J., Constantine, R., Crawford, R.J.M., Dalla Rosa, L., de Bruyn, P.J.N., Delord, K., Descamps, S., Double, M., Emmerson, L., Fedak, M., Friedlaender, A., Gales, N., Goebel, M., Goetz, K.T., Guinet, C., Goldsworthy, S.D., Harcourt, R., Hinke, J.T., Jerosch, K., Kato, A., Kerry, K.R., Kirkwood, R., Kooyman, G.L., Kovacs, K.M., Lawton, K., Lowther, A.D., Lydersen, C., Lyver, P., Makhado, A.B., Márquez, M.E.I., McDonald, B.I., McMahon, C.R., Muelbert, M., Nachtsheim, D., Nicholls, K.W., Nordøy, E.S., Olmastroni, S., Phillips, R.A., Pistorius, P., Plötz, J., Pütz, K., Ratcliffe, N., Ryan, P.G., Santos, M., Southwell, C., Staniland, I., Takahashi, A., Tarroux, A., Trivelpiece, W., Wakefield, E., Weimerskirch, H., Wienecke, B., Xavier, J.C., Raymond, B. and Hindell, M.A. 2020. ‘The retrospective analysis of Antarctic tracking data project’. Scientific Data, 7: 94. DOI: https://doi.org/10.1038/s41597-020-0406-x
Santora, J. and Brown, E.T. 2010. ‘Spatial Distribution Patterns of Southern Bottlenose Whales, Hyperoodon planifrons, near the South Shetland Islands, Antarctica’. Marine Mammal Science, 26(4):960–968.
Santora, J.A. 2012. ‘Habitat Use of Hourglass Dolphins near the South Shetland Islands, Antarctica’. Polar Biology, 35:801-806. doi:10.1007/s00300-011-1133-8
Santora, J.A. and Veit, R.R. 2013. ‘Spatio-Temporal Persistence of Top Predator Hotspots near the Antarctic Peninsula’. Marine Ecology Progress Series, 487:287–304.
Santora, J.A., Schroeder, I.D. and Loeb, V.J. 2014. ‘Spatial Assessment of Fin Whale Hotspots and Their Association with Krill Within an Important Antarctic Feeding and Fishing Ground’. Marine Biology, 161:2293–2305.
Schwarz, L.K., Goebel, M.E., Costa, D.P., Kilpatrick, A.M. 2013. ‘Top‐down and bottom‐up influences on demographic rates of Antarctic fur seals Arctocephalus gazelle’. Journal of Animal Ecology, 82:903– 911.
Secchi, E.R., Dalla Rosa, L., Kinas, P.G., Santos, M.C.P., Zerbini, A.N., Bassoi, M. and Moreno, I.B. 2001. ‘Encounter Rates of Whales Around the Antarctic Peninsula with Special Reference to Humpback Whales, Megaptera novaeangliae, in the Gerlache Strait: 1997/98 to 1999/2000’. Memoirs of the Queensland Museum, 47(2):571-578.
Secchi, E.R., Dalla Rosa, L., Kinas, P.G., Nicolette, R.F., Rufino, A.M.N. and Azevedo, A.F. 2011. ‘Encounter Rates and Abundance of Humpback Whales (Megaptera novaeangliae) in Gerlache and Bransfield Straits, Antarctic Peninsula’. Journal Cetacean Research and Management, (Special Issue)3:107–111.
Seyboth, E. 2018. ‘Ecologia Trófica e Distribuição de Cetáceos na Península Antártica Ocidental’. PhD thesis. Rio Grande: Universidade Federal do Rio Grande – FURG.
Seyboth, E., Groch, K.R., Dalla Rosa, L., Reid, K., Flores, P.A.C. and Secchi, E.R. 2016. ‘Southern Right Whale (Eubalaena australis) Reproductive Success is Influenced by Krill (Euphausia superba) Density and Climate’. Scientific Reports, 6:28205.
Siniff, D.B. and Stone, S. 1985. ‘The Role of the Leopard Seal in the Tropho-Dynamics of the Antarctic Marine Ecosystem’. In: W.R. Siegfried, P.R. Condy and R.M. Laws (eds.) Antarctic Nutrient Cycles and Food Webs. pp. 555-560. Berlin, Heidelberg: Springer.
Siniff, D.B., Garrott, R.A., Rotella, J.J., Fraser, W.R. and Ainley, D.G. 2008. ‘Projecting the Effects of Environmental Change on Antarctic Seals’. Antarctic Science, 20(5):425-435. DOI: 10.1017/S0954102008001351
Southwell, C., Bengtson, J., Bester, M.N., Schytte-Blix, A., Bornemann, H., Boveng, P. and Plötz, J. 2012. ‘A Review of Data on Abundance, Trends in Abundance, Habitat Utilisation and Diet for Southern Ocean Ice-Breeding Seals’. CCAMLR Science, 19:1-49.
Stammerjohn, S., Massom, R., Rind, D. and Martinson, D. 2012. ‘Regions of Rapid Sea Ice Change: An Inter-Hemispheric Seasonal Comparison’. Geophysical Research Letters, 39:L06501.
Staniland, I.J., Reid, K. and Boyd, I.L. 2004. ‘Comparing individual and spatial influences on foraging behaviour in Antarctic fur seals Arctocephalus gazella’. Marine Ecology Progress Series, 275:263-274.
Staniland, I.J. and Robinson, S.L. 2008. ‘Segregation between the sexes: Antarctic fur seals, Arctocephalus gazella, foraging at South Georgia’. Animal behaviour, 75(4):1581-1590.
Staniland, I.J., Morton, A., Robinson, S.L., Malone, D. and Forcada, J. 2011. ‘Foraging behaviour in two Antarctic fur seal colonies with differing population recoveries’. Marine Ecology Progress Series, 434:183-196.
Staniland, I.J., Robinson, S.L., Silk, J.R.D., Warren, N. and Trathan, P.N. 2012. ‘Winter distribution and haul-out behaviour of female Antarctic fur seals from South Georgia’. Marine Biology, 159(2):291-301.
Stevick, P., Aguayo-Lobo, A., Allen, J., Avila, I.C., Capella, J., Castro, C., Chater, K., Dalla Rosa, L., Engel, M.H., FéLix, F., Flórez-González, L., Freitas, A., Haase, B., Llano, M., Lodi, L., Muñoz, E., Olavarría, C., Secchi, E., Scheidat, M. and Siciliano, S. 2004. ‘Migrations of Individually Identified Humpback Whales Between the Antarctic Peninsula and South America’. Journal of Cetacean Research and Management, 6:109–113.
Stone, G.S. and Hamner, W.H. 1988. ‘Humpback Whales, Megaptera novaeangliae, and Southern Right Whales, Eubalaena australis, in Gerlache Strait, Antarctic’. Polar Record, 24(148):15-20.
Thiele, D., Chestera, E.T., Moore, S.E., Sirovic, A., Hildebrand, J.A. and Friedlaender, A.S. 2004. ‘Seasonal Variability in Whale Encounters in the Western Antarctic Peninsula’. Deep-Sea Research II, 51:2311-2325.
Turner, J., Lu, H., White, I., King, J., Phillips, T., Hosking, J., Bracegirdle, T., Marshall, G., Mulvaney, R. and Deb, P. 2016. ‘Absence of 21st century warming on Antarctic Peninsula consistent with natural variability’. Nature, 535:411-415.
Viquerat, S. and Herr, H. 2017. Mid-Summer Abundance Estimates of Fin Whales (Balaenoptera physalus) Around the South Orkney Islands and Elephant Island’. Endangered Species Research, 32:515–524. DOI: https://doi.org/10.3354/esr00832.
Weinstein, B.G. and Friedlaender, A.R. 2017. ‘Dynamic Foraging of a Top Predator in a Seasonal Polar Marine Environment’. Oecologia, 185:427-435.