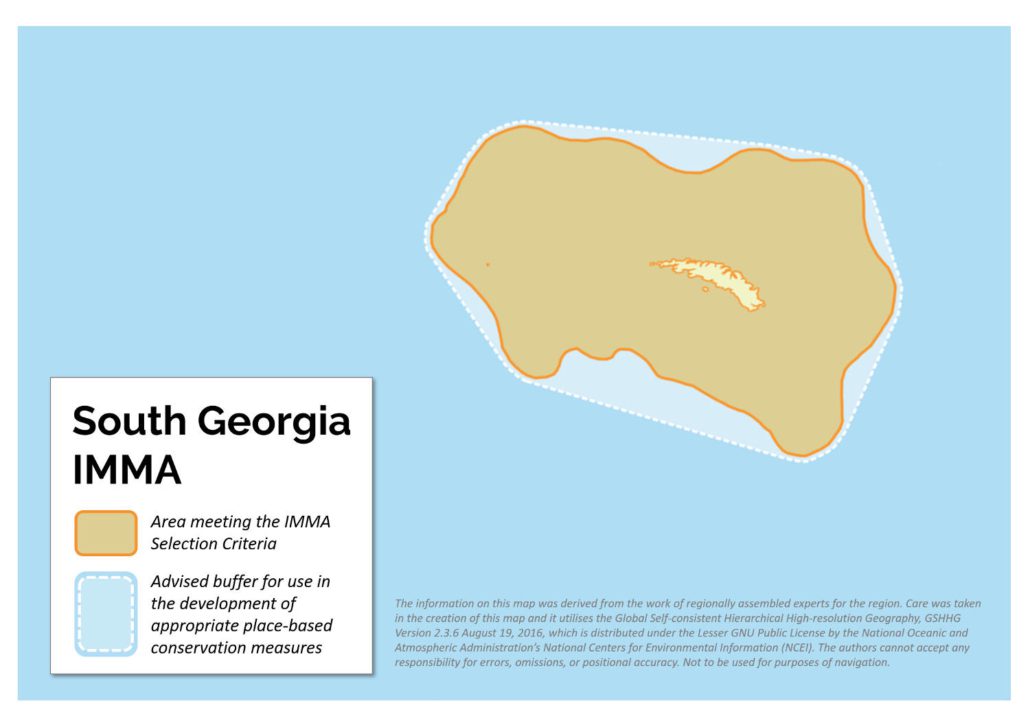
Size in Square Kilometres
423425
Qualifying Species and Criteria
Antarctic fur seal – Arctocephalus gazella
Criterion B (2); C (1; 2)
Weddell seal – Leptonychotes weddellii
Criterion C (1); D (1)
Southern elephant seal – Mirounga leonina
Criterion B (2); C (1)
Criterion D (2) – Marine Mammal Diversity
Other Marine Mammal Species Documented
Hydrurga leptonyx
Summary
South Georgia is a large subantarctic island, surrounded by a broad continental shelf, lying at the northeast corner of the Scotia Arc. The region is an area of high krill (Euphausia superba) density that sustains diverse marine mammal and other large predator populations. Ninety-five percent of the world’s population of Antarctic fur seals (Arctocephalus gazella) breeds on South Georgia. During the breeding season, lactating female seals are restricted in their foraging distribution by the need to return to land to feed their dependent pups. The island’s beaches are also important breeding and moulting areas for southern elephant seals (Mirounga leonina) representing over 50% of the world’s pup production. There is a small declining population of Weddell seals (Leptonychotes weddellii) at Larsen Harbour on the southeastern end of the island that is unique in that they are the only colony of this species that breeds away from the Antarctic continent, and to pup on land rather than on sea-ice. For details on the cetaceans that occur within this region, please see the summary for the Scotia Arc IMMA.
Description of Qualifying Criteria
Criterion A – Species or Population Vulnerability
Criterion B – Distribution and Abundance
Sub-criterion B1 – Small and Resident Populations
Sub-criterion B2 – Aggregations
Adult southern elephant seals (Mirounga leonina) return to land in summer (Jan – Feb) to moult for around one month (Carrick et al. 1962). Old skin and hair are replaced in a concentrated period during which elephant seals remain on land to avoid the cold water. The beaches of South Georgia are prime sites for elephant seals during this period when they form large aggregations (Le Boeuf & Laws 1994).
Criterion C: Key Life Cycle Activities
Sub-criterion C1 – Reproductive Areas
There is no recent complete worldwide estimate of southern elephant seal abundance although it is estimated that there were some 650,000 in the mid1990s (SCAR EGS 2008). Four distinct populations are recognised in the Southern Ocean (Gales et al. 1989, Hoelzel et al. 1993, Slade et al. 1998). While there is some movement of breeding individuals between these populations, it is rare (Fabiani et al. 2003, Reisinger and Bester 2010). On South Georgia, a census of southern elephant seal females in 1995 estimated a total of 113,444 individuals which suggested the population size was stable. There have been no subsequent counts in this region. This represents over 50% of the world pup production (Boyd 1996). Elephant seals are considered capital breeders in that females provision their young using stored fat reserves and do not go to sea to feed during lactation. During the breeding season adult females typically spend a month ashore (September-October), while adult males can fast on land for one to three months (August – October). Females give birth to a single pup 3 – 7 days after their arrival, and then suckle for around three weeks. Mating takes place shortly before the pup is weaned and the adult female returns to the sea a few days later. After weaning pups remain around the breeding beaches to moult their natal pelage before they depart to sea. Breeding aggregations are ephemeral, and do not last beyond the breeding season.
The last published census of Antarctic fur seals (Arctocephalus gazella) at South Georgia was in 1991/92, when an estimated 269,000 pups were counted (Boyd 2004). However, this was, a year of low food availability and the total population of this site in 1999/2000 was estimated to be between 4.5 and 6.2 million (I. Boyd pers. comm. in SCAR EGS 2008). There is evidence from the Bird Island long-term monitoring site that there has been a decline of ~30% between 2003 and 2012 (Forcada & Hoffman 2014). Overall, the South Georgia population is considered to represent 95% of the world’s Antarctic fur seals. These seals breed on beaches predominately at the north-western end of the island with smaller colonies around Cooper bay, Cooper Island and Clerke rocks (Boyd 2004). Males come ashore in November and fight to hold territories. Females begin to arrive a few weeks later, typically peaking in numbers in the first two weeks of December. Within 1-2 days of their arrival females give birth and then suckle on the beaches for 7-10 days. Towards the end of this perinatal period, they come into oestrus and mating takes place (Forcada & Staniland 2018). By early January, when the majority of females have been mated, males vacate the island moving southwards to the pack-ice, Antarctic islands and the Peninsula. Female fur seals are income breeders in that they must alternate trips to sea to feed with periods ashore suckling their pups that remain ashore. Therefore, females and pups are dependant on the island and its surrounding waters during the four-month long lactation period. Pups are typically weaned in April after which females and juveniles disperse at sea, although many remain in the vicinity of the island (Boyd et al 2002, Warren et al. 2006, Staniland et al. 2012).
A breeding colony of Weddell seals (Leptonychotes weddellii) exists in and near Larsen Harbour at the south-eastern end of South Georgia. This is the most northerly breeding site of the species, which is separated by about 800 km from the nearest colony in the South Orkney Islands (Burton 2015). This colony is unique not only because of its isolation but in that fact that pupping takes place on snow-covered beaches rather than on ice as at all other sites in Antarctica (Vaughan 1968; Bonner 1985).
Sub-criterion C2: Feeding Areas
The central place foraging behaviour of the female fur seals during their four-month lactation period means they are restricted in how far they can travel from the breeding beaches between December and April. Tracking data from these animals at a number of representative breeding colonies around South Georgia shows that they exploit areas of the shelf, shelf break and nearby oceanic waters (up to ~350 from the colony, BAS unpublished, Boyd et al. 2002, Staniland et al. 2011, Staniland et al. 2004). Their diet in this region is dominated by Antarctic krill (Euphausia superba), but also contains fish (Myctophiids and Notothenids) and squid. Although male fur seals fast when holding territories, tracking data has shown that during the mating season, when they are resident in large numbers at South Georgia, they also act as central place foragers (Staniland and Robinson 2008). Although there is a degree of niche separation with males favouring the shallow shelf areas closer to the island. During the winter months a large proportion of the population is thought to remain in close to the island, as juveniles and adult males are routinely seen on the beaches, observed by fishing vessels and a significant proportion of tracked female seals are resident but do not typically come ashore (Staniland pers com., CCAMLR observer data, Staniland et al. 2012, Boyd 2002, Reid 1995).
Sub-criterion C3: Migration Routes
C3a – Whale Seasonal Migratory Route
C3b – Migration / Movement Area
Criterion D – Special Attributes
Sub-criterion D1 – Distinctiveness
A colony of Weddell seals breeds in and near Larsen Harbour at the south-eastern end of South Georgia. This is the most northerly breeding site of the species, which is separated by about 800 km from the nearest colony in the South Orkney Islands (Burton 2015). This colony is unique not only because of its isolation but in that fact that pupping takes place on snow-covered beaches rather than on ice as at all other sites in Antarctica (Vaughan 1968; Bonner 1985). Accurate estimates of pup production are impossible with the low frequency of visitation by humans to this site, but it appears that the population has decreased over the last three decades. There was an apparent increase in the mid-1980s with a peak count of 150 adults and 58 pups and then a subsequent decline in pup production that appears to have started in the 1990s and continues to the present date with an average annual pup count of 8-9 in this century.
Sub-criterion D2 – Diversity
Supporting Information
Atkinson, A., Whitehouse, M. J., Priddle, J., Cripps, G. C., Ward, P., & Brandon, M. A. 2001. South Georgia, Antarctica: a productive, cold water, pelagic ecosystem. Marine Ecology Progress Series, 216, 279-308.
Bonner, W. N. 1985. Birds and mammals—Antarctic seals. Antarctica, (4), 202.
Boyd, I. L. 1993. Pup production and distribution of breeding Antarctic fur seals (Arctocephalus gazella) at South Georgia. Antarctic Science, 5(1), 17-24.
Boyd, I. L., Walker, T. R., & Poncet, J. 1996. Status of southern elephant seals at South Georgia. Antarctic Science, 8(3), 237-244.
Boyd, I. L., Staniland, I. J., & Martin, A. R. 2002. Distribution of foraging by female Antarctic fur seals. Marine Ecology Progress Series, 242, 285-294.
Burton, R. 2015. The Weddell seals of Larsen Harbour, South Georgia: A unique but apparently declining colony. Polar Record, 51(6), 667-671. doi:10.1017/S0032247414000953
Gales, N.J., Adams, M. and Burton, H.R. 1989. Genetic relatedness of two populations of the southern elephant seal, Mirounga leonina. Marine Mammal Science 5: 57-67.
Forcada, J. and Hoffman, J.I. 2014. Climate change selects for heterozygosity in a declining fur seal population. Nature 511: 462-465.
Forcada, J. and Staniland I.J. 2018. Antarctic Fur Seal: Arctocephalus gazella. In Bernd Würsig, J.G.M. Thewissen, Kit M. Kovacs (eds), Encyclopedia of Marine Mammals (Third Edition), Pages 25-27,ISBN 9780128043271,https://doi.org/10.1016/B978-0-12-804327-1.00046-7.
Hoelzel, A. R., Halley, J., O’Brien, S. J., Campagna, C., Arnbom, T., Le Boeuf, B. J., Ralls, K. and Dover, G. A. 1993. Elephant seal genetic variation and the use of simulation models to investigate historical population bottlenecks. Journal of Heredity 84: 443-449.
Le Boeuf, B. J., & Laws, R. M. (Eds.). 1994. Elephant seals: population ecology, behavior, and physiology. Univ of California Press.
Reid, K. 1995. The diet of Antarctic fur seals (Arctocephalus gazella Peters 1875) during winter at South Georgia. Antarctic Science, 7(3), 241-249
SCAR-EGS. 2008. Scientific Committee for Antarctic Research – Expert Group on Seals Report. Available at: http://www.seals.scar.org/pdf/statusofstocs.pdf. (Accessed: 5 November 2014).
Slade, R. W., Moritz, C., Hoelzel, A. R., and Burton, H. R. 1998. Molecular population genetics of the southern elephant seal Mirounga leonina. Genetics 149: 1945-1957.
Staniland, I. J., Reid, K., & Boyd, I. L. 2004. Comparing individual and spatial influences on foraging behaviour in Antarctic fur seals Arctocephalus gazella. Marine Ecology Progress Series, 275, 263-274.
Staniland, I. J., & Robinson, S. L. 2008. Segregation between the sexes: Antarctic fur seals, Arctocephalus gazella, foraging at South Georgia. Animal Behaviour, 75(4), 1581-1590.
Staniland, I. J., Morton, A., Robinson, S. L., Malone, D., & Forcada, J. 2011. Foraging behaviour in two Antarctic fur seal colonies with differing population recoveries. Marine Ecology Progress Series, 434, 183-196.
Staniland, I. J., Robinson, S. L., Silk, J. R. D., Warren, N., & Trathan, P. N. 2012. Winter distribution and haul-out behaviour of female Antarctic fur seals from South Georgia. Marine Biology, 159(2), 291-301.
Staniland, I. J., Ratcliffe, N., Trathan, P. N., & Forcada, J. 2018. Long term movements and activity patterns of an Antarctic marine apex predator: The leopard seal. PloS one, 13(6), e0197767.
Vaughan, R. W. 1968. The status of the Weddell seal (Leptonychotes weddellii) at South Georgia. British Antarctic Survey Bulletin, 15, 71-74.
Warren, N. L., Trathan, P. N., Forcada, J., Fleming, A., & Jessopp, M. J. 2006. Distribution of post-weaning Antarctic fur seal Arctocephalus gazella pups at South Georgia. Polar Biology, 29(3), 179-188.
Downloads
Download the full account of the South Georgia IMMA using the Brochure button below:
To make a request to download the GIS Layer (geopackage and/or geojson) for the South Georgia IMMA please complete the following Contact Form: