Size in Square Kilometres
152890
Qualifying Species and Criteria
Amazon river dolphin – Inia geoffrensis
Criterion A; C (2)
West Indian Manatee – Trichechus manatus
Criterion A; C (1, 2); D1
Amazonian manatee – Trichechus inunguis
Criterion A; C (1); D1
Guiana dolphin – Sotalia guianensis
Criterion B (1); C (1)
Criterion D (2) – Marine Mammal Diversity
Delphinus delphis, Globicephala macrorhynchus, Grampus griseus, Inia geoffrensis, Lagenodelphis hosei, Lontra longicaudis, Megaptera novaeangliae, Pseudorca crassidens, Pteronura brasiliensis, Sotalia guianensis, Stenella longirostris, Steno bredanensis, Trichechus manatus manatus, Trichechus inunguis, Tursiops truncatus
Other Marine Mammal Species Documented
Summary
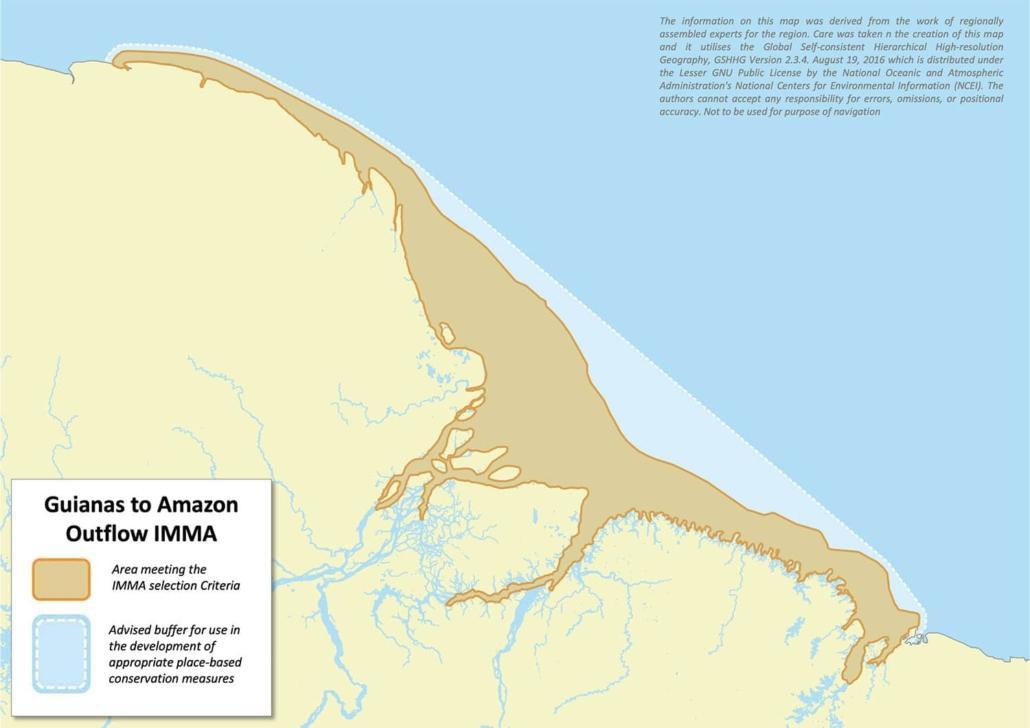
The Guianas to Amazon Outflow IMMA extends from central Guianas to Maranhão, Brazil overlapping with the Amazonian-Orinoco Influence Zone EBSA. A predominant feature of the IMMA is the outflow, the world’s largest river discharge, a low salinity (###lt##35ppth) plume with high concentrations of suspended material and nutrients spreading over thousands of kilometers along the North Brazilian Continental Shelf and the Equatorial Atlantic Ocean. This freshwater plume and its associated nutrients lead to increased productivity, creating an important feeding area for the survival of a rich local community of aquatic mammals. The composition of the IMMA’s cetacean community is more similar to those in the Caribbean than to cetaceans of the north-eastern Brazilian coast. The IMMA also encompasses the Great Amazon Reef System, which extends from French Guiana to Pará in Brazil. The IMMA provides habitat for several estuarine and coastal species that are threatened due to their restricted habitat requirements that overlap with human activity. These include the Guiana dolphin (Sotalia guianensis), the Amazon River dolphin (Inia geoffrensis,). These dolphins rely on mangrove ecosystems and estuaries, forming discrete populations with small home ranges. Additionally, the IMMA provides habitat for both the Amazonian manatee (Trichechus inunguis) and the West Indian manatee (Trichechus manatus), representing unique contact zone where hybrids between both species occur.
Description of Qualifying Criteria
Criterion A – Species or Population Vulnerability
The Guiana dolphin (Sotalia guianensis) has a discontinuous distribution along most of its range along the Atlantic coast of northern and eastern South America, and small resident populations can be found in distinct pockets of habitat in mangrove ecosystems, estuaries and bays (Bordin et al., 2022, Domit et al., 2021, Marmontel et al., 2021, Pool, 2019, Domit et al. 2021). Although the species is thought to occur along this entire range, Guiana dolphins usually form discrete populations, in which individuals typically have relatively small home ranges (Domit et al., 2021 and references therein). In 2018, the global IUCN Red list status of the Guiana dolphin was changed from Data Deficient to Near Threatened (Secchi et al. 2018). However, in French Guiana the Guiana dolphin is listed as Endangered (Moncorps et al., 2017) and in Brazil as Vulnerable (Secchi et al., 2018).
The Amazon Outflow IMMA is important for the survival and recovery of the Amazon River dolphin (Inia geoffrensis) which has been assessed as Endangered on the IUCN Red List of Threatened Species (Da Silva et al., 2018), and two species of manatees: the West Indian (Trichechus manatus) and the Amazonian manatee (T. inunguis) both classified as Vulnerable on the IUCN Red List (Deutsch et al. 2008; Marmontel et al. 2016). Moreover, the Amazon River plume has led to the expansion of the geographic distribution of the the Araguaian form of Inia geoffrensis, the only endemic cetacean of Brazil, into the shoreline of Marajó Bay and to the east, namely into the Curuçá Estuary, the eastern part of the Amazon Delta (Pará River) and approximately 500 km downstream of the Tucuruí Dam (Siciliano et al., 2016).
Some studies indicate that the West Indian manatee population in South America has declined over the years due to various anthropogenic factors (Lima et al., 2011; Alves et al., 2013; Alves et al., 2015). Meireles et al. (2022) evaluated the extinction risk of the West Indian manatee in Brazil. Although available data on the species’ abundance and mortality are not sufficient to apply the IUCN criteria, the authors worked with limited information following the guidelines, inferring, and projecting based on evidence to produce information and properly evaluate the West Indian manatee risk of extinction. The results indicated that T. manatus is Critically Endangered in Brazil due to the extremely high suspected and projected population reduction (=>80%) in the past and the future. Furthermore, although the causes of significant mortality were reduced in some parts of the species’ range, such as from hunting, other threats are increasing and are not expected to decrease in the coming decades.
Bonvicino (2020) established the presence of the Amazon manatee throughout the lower Amazon and its tributaries, including Marajó Island and its Atlantic coast and coastal islands, and from the northern (Amapá state, Brazil) to the southern shore of the Amazon estuaries (Pará state, Brazil). The current IMMA also hosts a hybridization zone of West Indian and Amazonian manatees. There are records of interspecific hybrids of T. inunguis and T. manatus close to the Amazon River mouth and along the Guiana Shield coastline (Noronha et al. 2022).
Criterion B – Distribution and Abundance
Sub-criterion B1 – Small and Resident Populations
The Guiana dolphin has a discontinuous distribution along most of its range and small resident populations are present year-round in small pockets in estuaries and bays (Bordin et al., 2022, Marmontel et al., 2021, Pool, 2019). These areas are used by these resident populations for feeding, mating, resting, and all biological functions related to reproduction, such as giving birth, nursing and caring for calves (Pool, 2019).
There are few abundance/density studies of Guiana dolphin distribution. Large scale aerial surveys conducted in 2008 and 2017 estimated Guiana dolphin abundance/density in French Guiana to be rouhgly 1,700 individuals (95% CI: 700-4 000; Laran et al, 2019; Mannocci et al, 2013). However, recently with coastal aerial surveys conducted in 2013, 2014 and 2019 concluded to lower estimates (Bordin et al, 2022).
Currently, the size of the population of Guiana dolphins in the Suriname River estuary is estimated to be between 80-100 based on photo identification. Of these, 67 are identified using dorsal fin identification and mark and recapture methodology, and some individuals have been resighted on multiple occasions since 2007 (Pool, 2019).
Sub-criterion B2 – Aggregations
Criterion C: Key Life Cycle Activities
Sub-criterion C1 – Reproductive Areas
The West Indian manatee and the Amazonian manatee occur in the Marajó Bay area and in the inlets and channels near Belém (under the influence of the Amazon River Plume). The Amazon Aquatic Mammal Study Group (GEMAM) coordinates a collaborative network to locate manatees trapped in fishing nets and conduct rescues along the coast of the state of Pará and in the interior of Belém. The presence of the Amazonian manatee is confirmed with the rescue and sightings of calves and adults in the great Belém area (Emin-Lima et al. 2021). On October 31st 2021, Amazonian manatees were sighted swimming near a beach in the Mosqueiro district, in greater Belém where mating behaviour was filmed by residents and tourists.
Both species of manatees are threatened by anthropogenic factors, such as habitat loss due to mangrove destruction, and bycatch in fishing gear (Parente et al., 2004; Borges et al., 2007; Meirelles, 2008; Silva et al., 2016). Strandings of neonates and calves of both species have been reported in the Amazon estuary, Tocantis Bay and Oiapoque estuary, all important breeding sites for both, West Indian and Amazonian manatees (Parente et al., 2004; Borges et al., 2007; Meirelles, 2008; Silva 2015).
Sub-criterion C2: Feeding Areas
The discharge of the Amazon River plume promotes a change in the freshwater layer and consequently in local productivity (Tosseto et al., 2022), creating an important feeding area for all coastal species, including Guiana dolphins and Amazonian manatees, as well as other aquatic vertebrates, such as fishes and sea birds (Siciliano et al. 2008; Costa et al. 2017; Costa et al. 2020; Tosseto et al., 2022).
The coastal waters of the IMMA are largely influenced by the massive suspended-sediment discharge, causing a mega-turbidity on the shelf, a growth of a subaqueous delta and an intermediary salinity (Tosseto et al., 2022). All these factors have a direct influence on oceanic biodiversity, including marine species, coral reef endemics and threatened species completely dependent on ecosystems such as estuaries, mudflats, sandy beaches and, particularly, littoral mangroves (Artigas et al. 2003).
A study of bone and teeth samples collected from museum samples and stranded or floating aquatic mammal carcasses in the IMMA Amazon estuarine complex and adjacent coastal zone and along Northeastern coast of Brazil included 220 samples of Guiana dolphins, and 11 from Amazonian manatees. Stable isotope analysis to analyse trophic relationships in the IMMA revealed that both species were feeding within the IMMA, and that there was clear resource partitioning between Guiana dolphins and Amazonian manatees. Isotopic niches were highly distinct, with no overlap among these two species inhabiting the Amazone plume. The study also indicated that both species had a fairly wide variety in their prey base and their ‘isotopic niche’, suggesting high habitat plasticity (Costa et al. 2020).
Sub-criterion C3: Migration Routes
C3a – Whale Seasonal Migratory Route
C3b – Migration / Movement Area
Criterion D – Special Attributes
Sub-criterion D1 – Distinctiveness
The West Indian (Trichechus manatus) and Amazonian (T. inunguis) manatee have an overlapping distribution at the mouth of the Amazon River. A result of this sympatry is the occurrence of hybrids, which are frequently found along the coasts of Amapá state in Brazil, French Guiana and Guyana. New genetic evidence (Lima et al. 2019) indicates the occurrence of a concentration of hybrids along the Guianas Shield coastline, which is an interspecific hybrid zone that also separates T. manatus populations located east (Brazil) and west (Caribbean, Gulf of Mexico, Florida and Antilles). In addition, Lima et al. (2019) suggest that this hybrid population, which occupies a unique mangrove-rich environment under strong influence of the Amazon River plume, requires independent conservation management measures.
The presence of hybrid manatees in this area under influence of the Amazon plume seems to be correlated both to the isolation of the western and eastern Trichechus m. manatus and the sympatric area between T. inunguis and T. m.manatus specimens (Vianna et al. 2006, Santos et al. 2016, de Oliveira et al. 2022).
Sub-criterion D2 – Diversity
The Amazon Outflow encompasses the Amazon River plume, including estuarine, coastal and oceanic zones. According to Siciliano et al. (2008) and Costa et al. (2017) local aquatic mammal richness ranged from 15 to 22 species, and the most common species are Guiana dolphins, Amazon river dolphins, and West Indian manatees (Siciliano et al. 2008; Santos et al. 2016, Costa et al. 2017). Common bottlenose dolphins (Tursiops truncatus) are also common throughout the IMMA (Mannocci, 2013; Pelagis, 2021). Additional species encountered during vessel-based and aerial surveys of the coastal areas of the IMMA include humpback whales (Megaptera novaeangliae), short-finned pilot whales (Globicephala macrorhynchus), false killer whales (Pseudorca crassidens), Risso’s dolphins (Grampus griseus), rough-toothed dolphins (Steno bredanensis), Fraser’s dolphins (Lagenodelphis hosei), common dolphins (Delphinus delphis), an d spinner dolphins (Stenella longirostris) (Ramos et al. 2001, Costa et al. 2017, Pelagis 2021, https://www.obsenmer.org/maps, Pusineri et al, 2021, De Boer, 2022). In addition, two species of otters are encountered in the area – giant otters (Pteronura brasiliensis) (e.g. Carter and Rosas, 2008, Duplaix et al. 2018) and neotropical otters (Lontra longicaudis) (e.g. Lopes Rheingantz, 2014).
Supporting Information
Abreu, C.T., 2015. Brazilian Coastal and Marine Protected Areas Importance, Current Status and Recommendations. University of Rhode Island.
Agência Nacional de Águas (Brasil) (ANA) (2015). HidroWeb: Sistemas de informações hidrológicas. Disponível em: http://hidroweb.ana.gov.br/HidroWeb. Accessed 22 January 2015.
Alves, M. D., Kinas, P. G., Marmontel, M., Borges, J. C. G., Costa, A. F., Schiel, N., & Aru ́jo, M. E. (2015). First abundance estimate of the Antillean manatee (Trichechus manatus manatus) in Brazil by aerial survey. Journal of the Marine Biological Association of the United Kingdom, 1, 1–12.
Alves, M.D.O., Schwamborn, R., Borges, J.C.G., Marmontel, M., Costa, A.F., Schettini, C.A.F., Araújo, M.E., 2013. Aerial survey of manatees, dolphins and sea turtles off northeastern Brazil: Correlations with coastal features and human activities. Biol. Conserv. 161, 91–100. doi:10.1016/j.biocon.2013.02.015
Artigas L.F., Vendeville P., Leopold M., Guiral D. & Ternon J-F. (2003). Marine biodiversity in French Guiana: estuarine, coastal, and shelf ecosystems under the influence of Amazonian waters. Gayana 67: 302-326.
Balensiefer, D.C., Attademo, F.L.N., Sousa, G.P., Freire, A.C. da B., da Cunha, F.A.G.C., Alencar, A.E.B., Silva, F.J. de L., Luna, F. de O., 2017. Three Decades of Antillean Manatee (Trichechus manatus manatus) Stranding Along the Brazilian Coast. Tropical Conservation Science 10, 194008291772837. https://doi.org/10.1177/1940082917728375
Barroso, C. X., Lotufo, T. M. C. & Matthews-Cascon, H. (2016). Biogeography of Brazilian prosobranch gastropods and their Atlantic relationships. J. Biogeogr. 43, 2477–2488.
Bezerra M.O., Medeiros C., Krelling A.P.M., Rosário R.P. & Rollnic M. (2011). Physical oceanographic behavior at the Guamá/Acará-Mojú and the Paracauari river mouths, Amazon coast (Brazil). In: Proceedings of the 11th international coastal symposium, Szczecin. Journal of Coastal Research 2011: 1448–1452
Bonvicino, C.R., Viana, M.C., Oliveira, E.H.C. de, Emin, R., Silva Junior, J.D.S. e, Sousa, M.E.M. de, Siciliano, S., 2020. Distribution of South American manatees, Trichechus manatus Linnaeus, 1758 and T. inunguis (Natterer, 1883) (Sirenia: Trichechidae). bcnaturais 15, 573–599. https://doi.org/10.46357/bcnaturais.v15i3.246
Bordin, A., Vanhoucke, M., Pineau, K., Kelle, L., Cozannet, N., Pool, M., Bolaños-Jiménez, J., De Thoisy, B., 2022. Study and conservation of the Guiana Dolphin (Sotalia guianensis) (Van Bénéden, 1864) in French Guiana. Lat. Am. J. Aquat. Mamm. https://doi.org/10.5597/lajam00276
Borges, J.C.G., Vergara-Parente, J.E., Alvite, C.M. de C., Marcondes, M.C.C., Lima, R.P. de, 2007. Embarcações motorizadas: uma ameaça aos peixes-boi marinhos (Trichechus manatus) no Brasil. Biota Neotrop. 7, 199–204. https://doi.org/10.1590/S1676-06032007000300021
Borges, J.C.G., Araújo, P.G., Anzolin, D.G., Miranda, G.E.C., 2008. Identificação de itens alimentares constituintes da dieta dos peixes-boi marinhos (Trichechus manatus) na região nordeste do Brasil. Biotemas. 21 (2), 77–81.
Brandini F.P., Lopes R.M., Gutseit K.S. & Sassi R. (2007). Planctonologia na Plataforma Continental do Brasil: Diagnose e Revisão Bibliográfca. (CEMAR/MMA/CIRM/FEMAR, 1997).
Carter, S.K. and Rosas, F.C.W. (1997), Biology and conservation of the Giant Otter Pteronura brasiliensis. Mammal Review, 27: 1-26. https://doi.org/10.1111/j.1365-2907.1997.tb00370.x
Chollett I., Mumby P.J., Müller-Karger F.E. & Hu C. (2012). Physical environments of the Caribbean Sea. Limnol. Oceanogr. 57, 1233–1244.
Costa M.S. (2013). Aporte hídrico e do material particulado em suspensão para a baía do Marajó: influência dos rios Amazonas e Tocantins. Master Thesis, Universidade Federal do Pará.
Costa A.F., Siciliano S., Emin-Lima R., Martins B.M.L., Sousa M.E.M., Giarrizzo T., Silva Júnior J.S. (2017). Stranding survey as a framework to investigate rare cetacean records of the north and north-eastern Brazilian coasts. ZooKeys 688: 111–134. https://doi.org/10.3897/zookeys.688.12636
Costa, A.F., Botta, S., Siciliano, S. et al. (2020). Resource partitioning among stranded aquatic mammals from Amazon and Northeastern coast of Brazil revealed through carbon and nitrogen stable isotopes. Sci Rep 10, 12897. https://doi.org/10.1038/s41598-020-69516-8
da Silva, V., Trujillo, F., Martin, A., Zerbini, A.N., Crespo, E., Aliaga-Rossel, E. & Reeves, R. 2018. Inia geoffrensis. The IUCN Red List of Threatened Species 2018: e.T10831A50358152. https://dx.doi.org/10.2305/IUCN.UK.2018-2.RLTS.T10831A50358152.en. Accessed on 07 June 2023.
Duplaix, N., Evangelista, E. and Rosas, F.C.W. (2015) Advances in the study of giant otter (Pteronura brasiliensis) ecology, behavior, and conservation: a review. Latin American Journal of Aquatic Mammals 10(2): 75-98. http://dx.doi.org/10.5597/lajam00200
de Mahiques, M.M., Siegle, E., Francini-Filho, R.B., Thompson, F.L., de Rezende, C.E., Gomes, J.D., Asp, N.E., 2019. Insights on the evolution of the living Great Amazon Reef System, equatorial West Atlantic. Sci Rep 9, 13699. https://doi.org/10.1038/s41598-019-50245-6
de Oliveira EHC, Gomes AJB, Costa AF, Emin-Lima R, Bonvicino CR, Viana MC, Reis LMA, Vidal MD, Cavalcanti MVG, Attademo FLN, Luna FO, Siciliano S. 2022. Karyotypical confirmation of natural hybridization between two manatee species, Trichechus manatus and Trichechus inunguis. Life (Basel). 2022 Apr 20;12(5):616. doi: 10.3390/life12050616. PMID: 35629284; PMCID: PMC9145575.
Lopes Rheingantz, M., Saraiva de Menezes, J.F. and de Thoisy, B. 2014. Defining Neotropical otter Lontra longicaudis distribution, conservation priorities, and ecological frontiers. Tropical Conservation Science Vol.7 (2): 214-229. Available online: www.tropicalconservationscience.org
Meirelles, A.C.O., 2008. Mortality of the Antillean manatee, Trichechus manatus manatus, in Ceará State, north-eastern Brazil. J. Mar. Biol. Ass. 88, 1133–1137. https://doi.org/10.1017/S0025315408000817
Meirelles, A.C.O., Carvalho, V.L., Marmontel, M., 2018. West Indian Manatee Trichechus manatus in South America: Distribution, Ecology and Health Assessment, in: Rossi-Santos, M.R., Finkl, C.W. (Eds.), Advances in Marine Vertebrate Research in Latin America, Coastal Research Library. Springer International Publishing, Cham, pp. 263–291. https://doi.org/10.1007/978-3-319-56985-7_11
Meirelles, A.C.O., Lima, D. dos S., Alves, M.D. de O., Borges, J.C.G., Marmontel, M., Carvalho, V.L., Santos, F.R. dos, 2022. Don’t let me down: West Indian manatee, Trichechus manatus, is still critically endangered in Brazil. Journal for Nature Conservation 67, 126169. https://doi.org/10.1016/j.jnc.2022.126169
Oliveira, E.H.C., Gomes A.J.B., Costa A.F., Emin-Lima R., Bonvicino C.R., Viana M.C., Reis L.M.A., Vidal M.D., Cavalcanti M.V.G., Attademo F.L.N., Luna F.O. & Siciliano S. (2022). Karyotypical confirmation of natural hybridization between two manatee species, Trichechus manatus and Trichechus inunguis. Life 12, 5: 616. https://doi.org/10.3390/life12050616
Deutsch, C. J., Self-Sullivan, C., & Mignucci-Giannoni, A. (2008). Trichechus manatus: The IUCN Red List of Threatened Species 2008: e.T22103A9356917 [Data set]. https://doi.org/10.2305/IUCN.UK.2008.RLTS.T22103A9356917.en. Accessed on 20 May 2023.
Dos Santos, S. S.; Medeiros, I. S.; Rebelo, V. A.; Carvalho, A. O. B.; Dubut, J. P.; Mantovani, J. E.; Círiaco, R. D.; Dos Santos, R. E. G.; Marmontel, M.; Normande, I. C.; Velôso, T. M. G.; Borges, J. C. G. 2022. Home ranges of released West Indian manatees in Brazil. ORYX, 56 : 1-8.
Domit, C., de Miranda, A.V., Azevedo, A., Costa, A.F., de Meirelles, C.O., Farro, A.P., Bracarense, A.P.L., Barreto, A.S., Zaccaron, S., Andriolo, A., Louzada, C.N., Bertozzi, C.P., Marcondes, D.S., Ruenes, G., Melo-Santos, G., Cunha, H., Maciel, I., Jr, J.L.B., Torres-Florez, J.P., Tullio, J.D., Groch, K.R., May-Collado, L., Vidal, L.G., Flach, L., Sánchez, L., Londoño, M.C.R., Cremer, M.J., Alonso, M.B., Laeta, M., Cantor, M., Marmontel, M., Espinoza-Rodríguez, N., Fruet, P., Hauser, R., Sousa-Lima, R., Paitach, R., Tardin, R., Siciliano, S., Moura, S., Caballero, S., Bisi, T., Belleghem, T.V., Ridoux, V., Luz, V., Briceño, Y., Pendu, Y.L., 2021. Progress report of the Sotalia guianensis Intersessional Group: Status of the Current Knowledge and Action Plan 69.
Emin-Lima, R., Costa, A.F., Attademo, F.L.N., Hauser-Davis, R.A., Luna, F. de O., Siciliano, S., (2021). Amazonian Manatees (Trichechus inunguis) Inhabiting an Equatorial Metropolis: Historical Records and Mating Activity Near Belém, Northern Brazil = Sobre A Presença Do Peixe-Boi Amazônico (Trichechus Inunguis) Próximo À Belém: Registros Históricos E Atividade Reprodutiva. Bol. Lab. Hidrobiol. 31. https://doi.org/10.18764/1981-6421e2021.12
Emin-Lima, N.E. et al. (2022). Morphological disparity in the skull of Amazon River dolphins of the genus Inia (Cetacea, Iniidae) is inconsistent with a single taxon. Journal of Mammalogy, gyac039, https://doi.org/10.1093/jmammal/gyac039
Figueiredo, A.G., Allison, M., Nittrouer, C.A., 1991. Amazon discharge (Technical Report). Universidade Federal Fluminense, Niteroi.
Francini-Filho, R. B. et al. (2018). Perspectives on the Great Amazon Reef: Extension, biodiversity, and threats. Front. Mar. Sci. 5, 142.
Gozlan, R., Siblet, J.P., Moncorps, S., 2017. La liste rouge des espèces menacées en France : faune vertébrée de Guyane (No. fdi:010072525). UICN, Paris.
Hrbek, T., da Silva, V.M.F., Dutra, N., Gravena, W., Martin, A.R., Farias, I.P., 2014. A New Species of River Dolphin from Brazil or: How Little Do We Know Our Biodiversity. PLoS ONE 9, e83623. https://doi.org/10.1371/journal.pone.0083623
Jo, Y.-H., 2005. A study of the freshwater discharge from the Amazon River into the tropical Atlantic using multi-sensor data. Geophys. Res. Lett. 32, L02605. https://doi.org/10.1029/2004GL021840
Laran, S., Bassols, N., Dorémus, G., Authier, M., Ridoux, V., Van Canneyt, O., 2019. Distribution et abondance de la mégafaune marine aux Petites Antilles et en Guyane française. Campagne REMMOA – II. Rapport final pour l’Agence Française pour la Biodiversité. https://www.observatoire-pelagis.cnrs.fr/wp-content/uploads/2021/05/12-RAPPORT_REMMOA_ANTGUY_FINAL_2019.pdf
Lentz, S.J., Limeburner, R., 1995. The Amazon River Plume during AMASSEDS: Spatial characteristics and salinity variability. J. Geophys. Res. 100, 2355. https://doi.org/10.1029/94JC01411
Lima, C.S., Magalhães, R.F., Marmontel, M., Meirelles, A.C., Carvalho, V.L., Lavergne, A., Thoisy, B.D., Santos, F.R., 2019. A hybrid swarm of manatees along the Guianas coastline, a peculiar environment under the influence of the Amazon River plume. An. Acad. Bras. Ciênc. 91, e20190325. https://doi.org/10.1590/0001-3765201920190325
Lima, R. P., Paludo, D., Soavinski, R. J., Silva, K. G., Oliveira, E. M. A. 2011. Levantamento da distribuição, ocorrência e status de conservação do Peixe‐Boi Marinho (Trichechus manatus, Linnaeus, 1758) no litoral nordeste do Brasil. Natural Resources, 1: 41‐57.
Loder J. W., Boicourt W. C. & Simpson J.H. (1998). Western ocean boundary shelves coastal segment (W). Sea 11, 3–27.
Luna, F. de O., de Lima, R.P., de Araújo, J.P., 2008. Status de conservação do peixe-boi marinho (Trichechus manatus manatus Linnaeus, 1758) no Brasil 10.
Mannocci, L., Monestiez, P., Bolaños-Jiménez, J., Dorémus, G., Jeremie, S., Laran, S., Rinaldi, R., Van Canneyt, O., and Ridoux, V. (2013) Megavertebrate communities from two contrasting ecosystems in the western tropical Atlantic. Journal of Marine Systems 111–112, 208-222.
Marmontel, M., de Souza, D., & Kendall, S. (2016). Trichechus inunguis: The IUCN Red List of Threatened Species 2016: e.T22102A43793736 [Data set]. International Union for Conservation of Nature. https://doi.org/10.2305/IUCN.UK.2016-2.RLTS.T22102A43793736.en
Marmontel, M., Lima, D. dos S., Funi, C., Santos, V.F. dos, Oliveira-da-Costa, M., 2021. Unveiling the Conservation Status of Inia and Sotalia in the Brazilian Northeastern Amazon. Aquat Mamm 47, 376–393. https://doi.org/10.1578/AM.47.4.2021.376
Martins S.E.M. & Mendes A.C. (2011). Caracterização de depósitos sedimentares recentes da porção superior da Baía de Marajó (margem leste do estuário do Rio Pará, Amazônia). Pesquisas em Geociências 38(2): 168–180.
Meirelles A.C.O. (2008). Mortality of the Antillean manatee, Trichechus manatus manatus, in Ceará State, northeastern Brazil. Journal of the Marine Biological Association of the United Kingdom, v. 88, n.6, p. 1133–1137.
Moura, R. L. et al. (2016). An extensive reef system at the Amazon River mouth. Sci. Adv. 2, 1–11.
Muehe, D., Garcez, D.S., 2005. A Plataforma Continental Brasileira e Sua Relação com a Zona Costeira E A Pesca 20.
Neumann-Leitão, S. et al. (2018). Zooplankton from a reef system under the influence of the Amazon River plume. Front. Microbiol. 9, 1–15.
Nittrouer, C.A., DeMaster, D.J., 1996. The Amazon shelf setting: tropical, energetic, and influenced by a large river. Continental Shelf Research 16, 553–573. https://doi.org/10.1016/0278-4343(95)00069-0
Noronha R.C.R., Almeida B.R.R., Chagas M.C.S., Tavares F.S., Cardoso A.L., Bastos C.E.M.C., Silva N.K.N., Klautau A.G.C.M., Luna F.O., Attademo F.L.N., Lima D.S., Sabioni L.A., Sampaio M.I.C., Oliveira J.M., Nascimento L.A.S.D., Martins C., Vicari M.R., Nagamachi C.Y., Pieczarka J.C. (2022) Karyotypes of manatees: New insights into hybrid formation (Trichechus inunguis × Trichechus m. manatus) in the Amazon Estuary. Genes (Basel). 13(7):1263. doi: 10.3390/genes13071263. PMID: 35886048; PMCID: PMC9323068
Normande, I. C., Malhado, A. C. M., Reid, J., Viana, P. C., Savaget, P. V. S., Correia, R. A., Luna, F. O., Ladle, R. J. 2016. Post-release monitoring of Antillean manatees: an assessment of the Brazilian rehabilitation and release programme. Animal Conservation, 19: 235-246.
Pansard, K.C.A., Gurgel, H. de C.B., Andrade, L.C. de A., Yamamoto, M.E., 2011. Feeding ecology of the estuarine dolphin (Sotalia guianensis) on the coast of Rio Grande do Norte, Brazil. Marine Mammal Science 27, 673–687. https://doi.org/10.1111/j.1748-7692.2010.00436.x
Parente, C.L., Vergara-Parente, J.E., Lima, R.P., 2004. Strandings of Antillean manatees, Trichechus manatus manatus, in northeastern Brazil. Lat. Am. J. Aquat. Mamm. 3, 69–75. https://doi.org/10.5597/lajam00050
Pelagis (2021). Visual sightings of scientific surveys. https://pelabox.univ-lr.fr/pelagis/PelaObs/ Accessed May 2023.
Pool, M.S., 2019. Estuarine Mammal Baseline for the proposed Suriname River Dredging ESIA Update Project.
Prestes Y.O. (2013). Transporte de volume na zona de maré do rio Pará, Brasil. Monograph (Undergraduate), Institute of Geosciences, Universidade Federal do Pará.
Pusineri, C., Martinez, L., Bordin, A., Montgolfier, B., Deffes, O., Rufray, V., Lenrume, P., Safi, M., Suardi, A., Wyss, V., Geraldes, D., Marzelière, M., Leclerc, B., Barnicoat, S. and Rinaldi, R. (2021) Cetacean community off French Guiana. Latin American Journal of Aquatic Mammals 16(1): 12-22. https://doi.org/10.5597/lajam00266
Ramos et al. (2001). A note on strandings and age of sperm whales (Physeter macrocephalus) on the Brazilian coast. Journal of Cetacean Research and Management 3(3):321-327
Rocha, L. A. (2003). Patterns of distribution and processes of speciation in Brazilian reef fishes. J. Biogeogr. 30, 1161–1171.
Schwassmann H.O., Barthem R.B. & Carvalho M.L. (1989). A note on the seasonally shifting zone of high primary production in the Bay of Marajó, Pará, Brazil, 1983–1984. Acta Botanica Brasilica 2(1): 165–174.
Secchi, E., Santos, M.C. de O. & Reeves, R. 2018. Sotalia guianensis (errata version published in 2019). The IUCN Red List of Threatened Species 2018: e.T181359A144232542. https://dx.doi.org/10.2305/IUCN.UK.2018-2.RLTS.T181359A144232542.en. Accessed on 25 January 2023.
Siciliano S., Emin-lima N.R., Costa A.F., Tosi C.H., Garri R.G., Regina C., Silva D.A., Sousa J.D.E., Júnior E.S. (2008). Revisão do conhecimento sobre os mamíferos aquáticos da Costa Norte do Brasil. Arquivos do Museu Nacional 66: 381–401.
Siciliano, S., Valiati, V.H., Emin-Lima, R., Costa, A.F., Sartor, J., Dorneles, T., de Sousa e Silva Júnior, J., de Oliveira, L.R. (2016). New genetic data extend the range of river dolphins Inia in the Amazon Delta. Hydrobiologia 777, 255–269. https://doi.org/10.1007/s10750-016-2794-7
Siciliano, S. et al. 2023. Giving names to the characters: Identifying, tracing and estimating the multiple use of aquatic wildlife in Brazil. In: Galetti Jr., P.M. (eds) Conservation Genetics in the Neotropics. Springer, Cham. https://doi.org/10.1007/978-3-031-34854-9_13.
Silva, S.M. (2015). Caracterização populacional e estimativa de parentesco entre peixes-bois-marinhos Trichechus manatus (Linnaeus, 1758) usando marcadores microssatélites. Dissertação (Mestrado em Biologia Ambiental), Universidade Federal do Pará.
Silva CPN, Meirelles ACO, Umezaki J (2016) Ameaças. In: Meirelles ACO, Carvalho VL (eds) Peixe-boi-marinho: Biologia e Conservação no Brasil. São Paulo, Bambu, pp 93–108
Targino, A.K.G. & Gomes, P.B. (2020). Distribution of sea anemones in the Southwest Atlantic: Biogeographical patterns and environmental drivers. Mar. Biodivers. 50, 80.
Tosetto G.E., Bertrand, A., Neumann-Leitão, S. et al. (2022). The Amazon River plume, a barrier to animal dispersal in the Western Tropical Atlantic. Sci Rep 12, 537. https://doi.org/10.1038/s41598-021-04165-z
Downloads
Download the full account of the Guianas to Amazon Outflow IMMA using the Brochure button below:
To make a request to download the GIS Layer (geopackage and/or geojson) for the Guianas to Amazon Outflow IMMA please complete the following Contact Form: